Order Cut-Off & Processing
-
Order cut-off: 2:00 PM (PT).
-
Orders placed after 2:00 PM PT begin processing the next day.
-
Handling time: 1–3 days (Mon–Sun) to prepare your order for shipment.
Transit Time
Transit time: 3–7 days (Mon–Sun) once shipped.
Total delivery window: typically 4–10 days from order placement to delivery, depending on when the order is placed relative to cut-off, handling time, carrier performance, weather, and holidays.
Shipping Fee
Flat rate: $5.00 per order for standard shipping anywhere in the U.S.










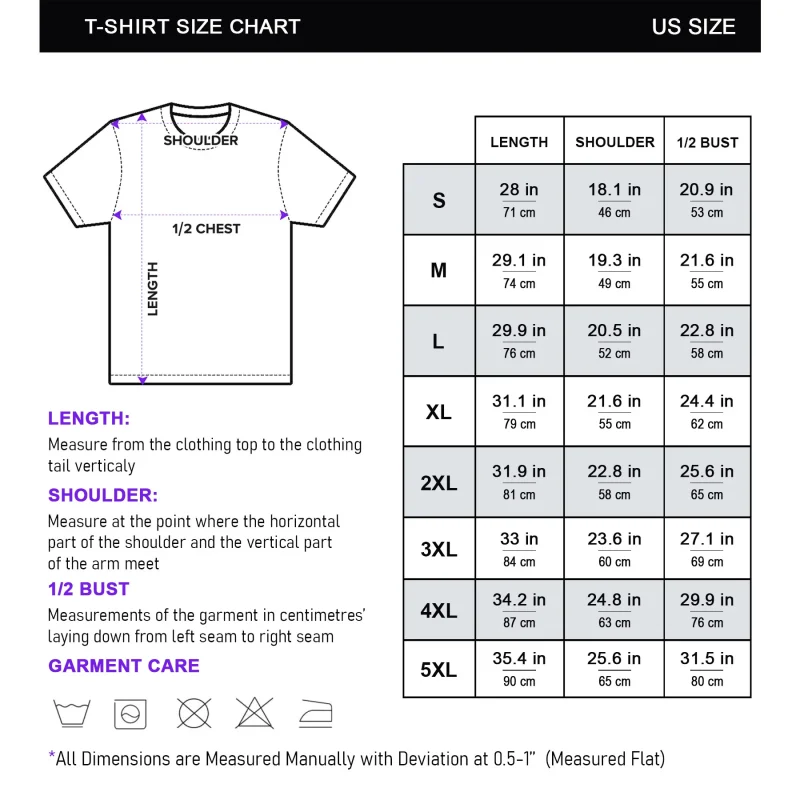
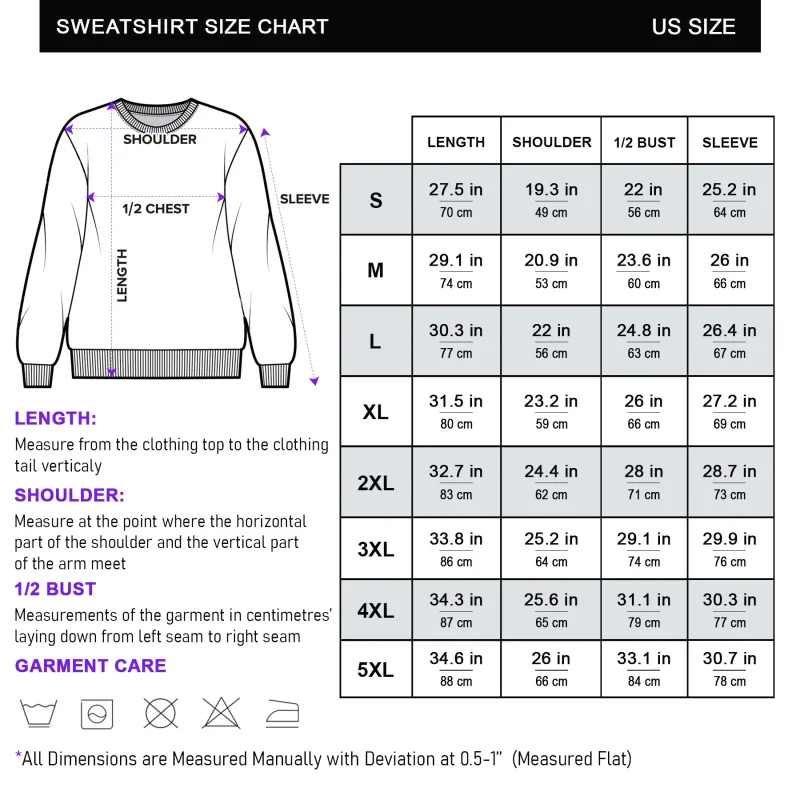
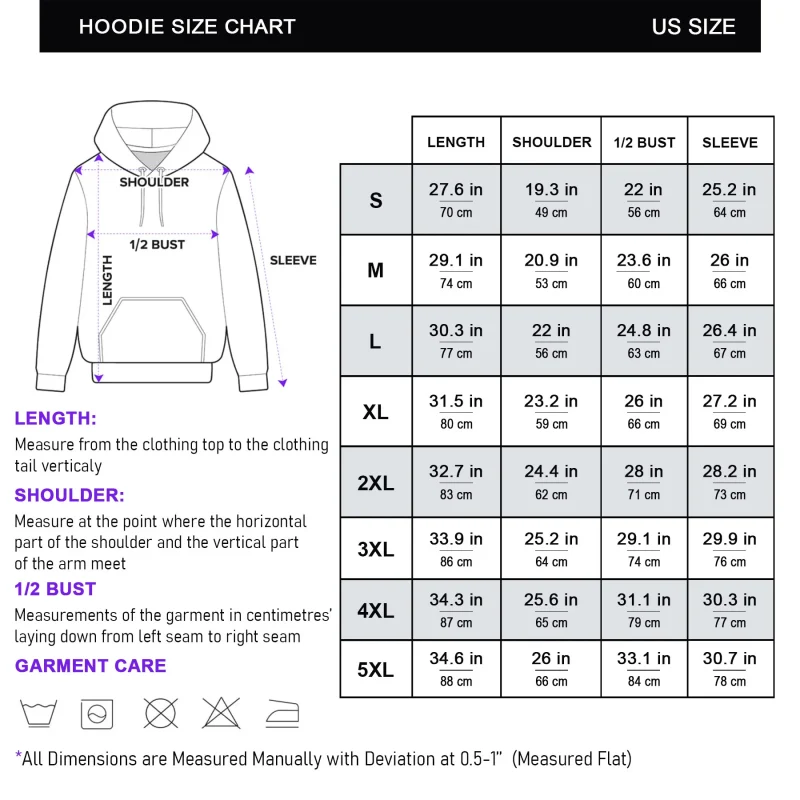
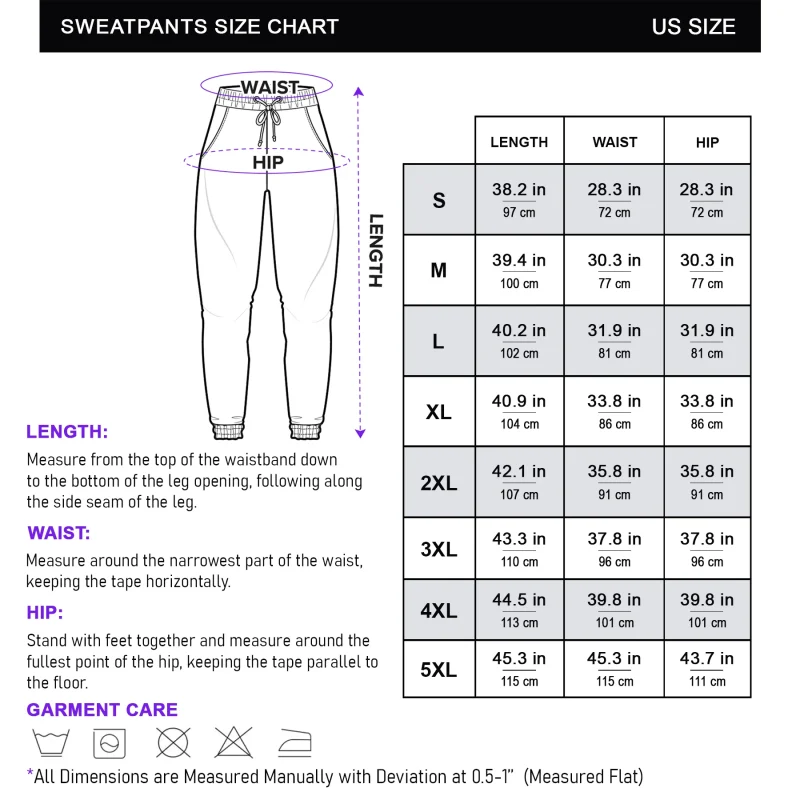
Reviews
There are no reviews yet.